 The Northern Ireland-based Authorised Representative or UK Responsible Person must provide the MHRA with details of the person placing the product on the Northern Ireland market if the person placing the product on the market is not: Please see below for further information.ĭistributors and suppliers are not required to register with the MHRA. Please note that new requirements have recently been introduced regarding the registration of custom-made devices in Northern Ireland. Where a Northern Ireland-based Authorised Representative is appointed, the Authorised Representative must register all devices with the MHRA. Where an EU-based Authorised Representative is appointed, the Great Britain-based manufacturer must register all device classes other than Class I devices and general IVDs with the MHRA. Great Britain manufacturers must designate an Authorised Representative based in the EU or Northern Ireland in order to place a device on the Northern Ireland market. 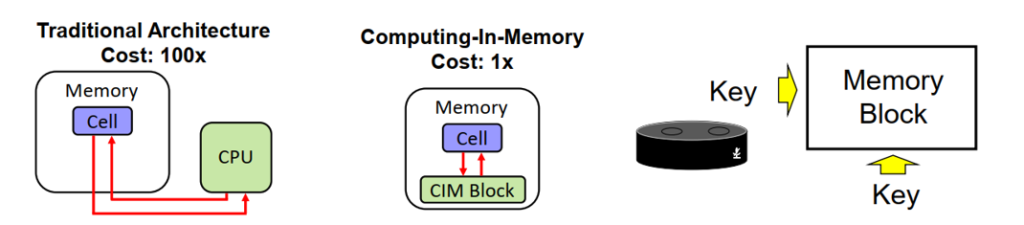
Requirements for Great Britain-based manufacturers placing devices on the Northern Ireland market See guidance for more information about the role of the UK Responsible Person. place IVDs on the Northern Ireland market that have not already been registered with an EU Competent Authority.have an Authorised Representative based outside Northern Ireland and.you are a manufacturer based outside the UK, the EU and the EEA and.Place IVDs on the Northern Ireland market that have not already been registered with an EU Competent Authority. 
you are a manufacturer based in the EU or the EEA and. 
The requirement to appoint a UK Responsible Person for the purposes of placing an IVD on the Northern Ireland market applies in cases where: Non-UK manufacturers are not required to appoint a UK Responsible Person for the purpose of placing general medical devices and active implantable medical devices on the Northern Ireland market. A UK Responsible Person is not required on order to place IVDs that do not fall within these device classifications on the Northern Ireland market. If you are a non-UK manufacturer, you may be required to appoint a single UK Responsible Person within the UK for IVD List A products, IVD List B products and self-test IVDs that are placed on the Northern Ireland market. Register your device to place on the Northern Ireland market Requirements for non-UK manufacturers Please see below for further information. The precise requirements depend on the location of the manufacturer, the location of the Authorised Representative and the device class. In some circumstances, it is a requirement of the UK MDR 2002 that you inform the MHRA when you first place your device on the Northern Ireland market. Placing a device on the Northern Ireland market In such cases, the UK Responsible Person or the manufacturer must provide the MHRA with the importer details, including their place of business in Great Britain. In cases where the Great Britain importer is not the UK Responsible Person, the importer must inform the relevant manufacturer or UK Responsible Person of their intention to import a device. Where any changes to registrations are made, a £100 statutory fee will apply per application.ĭistributors and suppliers are not required to register with the MHRA. The UK Responsible Person will then assume the responsibilities of the manufacturer in terms of registering the device with the MHRA. Please note that the accounts of any former Great Britain-based Authorised Representatives that have not updated their role to UK Responsible Person on the MHRA registration system, as well as the accounts of any represented manufacturers, will be suspended from 1 January 2022 until the UK Responsible Person has updated their role. If the manufacturer is based outside of the UK, they must appoint a single UK Responsible Person to take responsibility for all of their medical devices. Register your device to place on the Great Britain market
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
